News
Clinical Trials Offer Potential Treatments for Non-Alcoholic Fatty Liver Disease
July 30, 2019
University Gastroenterology & Hepatology Associates »
Four clinical trials at the UCSF Fresno Liver Program could help people who have scarring of the liver that did not result from alcohol abuse.
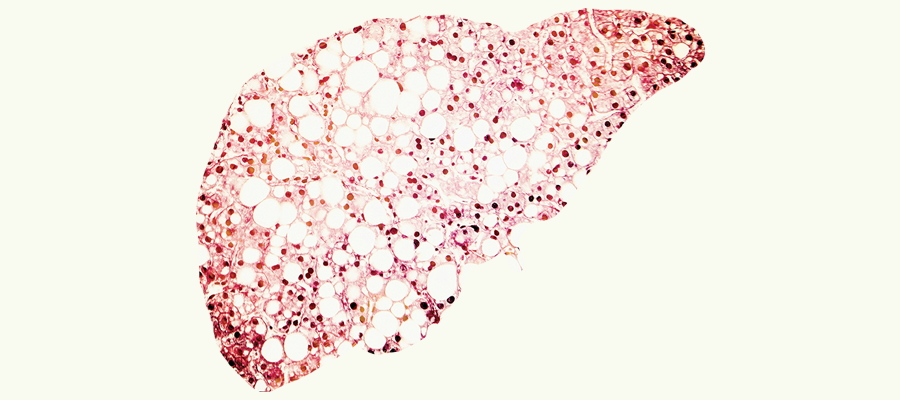
Scarring of the liver can be caused by several things, including chronic alcoholism, Hepatitis B or C, but for a growing number of people, the cause is an excessive accumulation of fat in the liver – a condition known as nonalcoholic fatty liver disease.
The double-blind placebo-controlled trials are part of multi-national trials to study the potential of medications for patients who have nonalcoholic fatty liver disease.
Approximately 100 million Americans have too much fat in their livers. In some people, there are no complications from fatty liver, but others develop nonalcoholic steatohepatitis (NASH), in which the fat in the liver triggers inflammation. Inflammation can damage healthy liver cells, causing scarring that affects the organ’s ability to function properly.
“Fatty liver disease is as serious as having a drinking problem,” said Marina Roytman, MD, FACP, and director of the UCSF Fresno Liver Program. “It can lead to cirrhosis of the liver and to liver cancer.”
Risk factors for fatty liver disease include diabetes, high blood pressure, high cholesterol, sedentary lifestyle, as well as being overweight or obese.
Obesity rates across the United States are increasing and so is the prevalence of fatty liver disease, including in Fresno County. According to the most recent data, 78.6 percent of Latino adults in Fresno County are overweight or obese; 64.9 percent of Caucasians are overweight or obese and 35.4 percent of Asian adults are overweight or obese.
Fatty liver disease can be reversed with weight loss, healthy eating and exercise, Roytman said. “The cool thing about the liver is, if we allow it, it can heal itself.” But changing eating and exercise habits is not an easy remedy for many people, and up to this point, there is no approved drug therapy for NASH patients, Roytman said.
The four clinical trials are aimed at reduction of steatosis (fat) and fibrosis (scarring) of the liver. The trials involve patients in varying stages of NASH, and target different points in disease development (from fat to inflammation to scarring). The scarring that causes progression to cirrhosis is the most important target, Roytman said.
The UCSF Fresno Liver Program is part of the Gastroenterology and Hepatology Fellowship. Fellows receive training in the diagnosis and management of all liver diseases.
